Scientific results (EN)
Executive summary of the activities carried out within the project
PN-III-P1-1.1-PD-2021-0552
Multicomponent hydrogel a hybrid structure obtained in situ by irradiation technology for malignant melanoma therapy (HYDRO-CAN)
CONTRACT NO. PD 20/2022
STAGE 1 – 2022
In the first stage of the project, 3 recipes of multicomponent hydrogels (labelled as PD81, PD9 and PD10) were synthesized by irradiation with electron beam (e-beam). Hydrogels were obtained from aqueous polymer mixtures in which different proportions of: bovine collagen gel, sodium carboxymethylcellulose, poly(vinylpyrrolidone), chitosan and poly(ethylene oxide) were used. The cross-linking reaction was carried out exclusively by irradiation at 25 kGy, a dose of irradiation sufficient both to complete the cross-linking reaction and also effective from the point of view of hydrogel sterilization. The obtained hydrogels were tested in terms of physical and chemical stability, mechanical, compositional, structural and morphological properties.
The hydrogels developed in this project are transparent, maintain their structure, are non-adhesive when handling and most importantly, especially from the application point of view, they have an elastic structure.
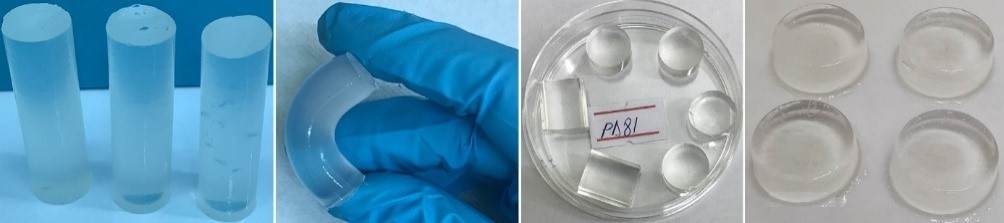 Figure 1. Appearance of hydrogels obtained after irradiation.
Figure 1. Appearance of hydrogels obtained after irradiation.
PD81, PD9 and PD10 hydrogels showed different degrees of swelling in deionized water and buffer solutions. The PD81 composition showed the highest degree of swelling (1100% - 3250% -1400%-1100%) in environments with pH 5.4-6.5-7.4-9.4, in these conditions the hydrogels are stable and do not dissociate in the swelling environment.
Hydrogels obtained by e-beam irradiation have rheological behavior characteristic of soft solids, they present a permanent macromolecular network predominantly elastic. PD81 and PD 9 hydrogels showed the highest G' values between 9697 Pa and 14, 980 Pa.
PD81 hydrogel showed the highest crosslinking density (Ve = 1.17 mol/m3), which shows the formation of a hydrogel with dense structure and high stability. The PD10 hydrogel showed a significant decrease in the crosslinking density, also correlated with the decrease in the elastic modulus. These results were suggestive for the selection of compositions PD81 and PD9 for further studies. Hydrogels retain between 84.8% and 89.2% of moisture for 12 hours, which provides a beneficial environment for much faster wound healing.
From a compositional point of view, from the analysis of the XPS spectra, peaks of significant intensity specific to C-C bonds (284.4 eV) were revealed, which suggests that the structure of the hydrogels is not degraded as a result of the irradiation process. In the case of PD10 hydrogel, a higher contribution of (C=O) and (O=C–O) bonds was observed, which was associated with degradative processes in the PD10 hydrogel structure.
Morphologically, PD81 and PD9 hydrogels show porous structure with interconnected and homogeneously distributed pores. The PD81 hydrogel presents in its structure, uniformly distributed micropores, this structure has better swelling capacity in environments with different pH, which also gives it a better loading capacity of this hydrogel, with drugs.
Testing the cytotoxic potential of the free drug (DOX) on melanoma cells from the A375 line in the concentration ranges 1.56 – 200 µg/ml and 0.01 – 1.56 µg/ml revealed that the IC50 dose is represented by the concentration range 0.01 µg/ml – 0.04 µg/ml.
In vitro evaluation of the biocompatibility potential of the developed hydrogels demonstrated that the highest biological performance as skin wound dressings is exhibited by samples PD81 and PD9, especially PD81, for which selected IC50 doses were loaded into the hydrogel PD81. These hydrogels were subsequently investigated for their biocompatibility with dermal fibroblast (CCD 1070SK) and epithelial cell (VERO) cell lines demonstrating that they are tolerated by normal cells.
STAGE 2 – 2023
Act. 2.1 consists of the determination of the biodegradation profiles and the in vitro release mechanisms in a dynamic regime of the active substances at physiological pH, infected wound, and tumor environment. Quantification of the maximum loading and release concentration of bioactive molecules (DOX) with anti-tumor effect was also pursued.
In the first 50 days, the percentage of biodegradation was only 0.04 %. The low biodegradation rate of PD81 hydrogel may be associated with a permanently cross-linked network formed upon irradiation. In a physiological environment, after 84 days, the biodegradation rate increased slightly, and the hydrogel lost only 0.10% of its initial mass, which suggests that the tested hydrogel has a stable structure. Quantification of the amount of DOX loaded and respectively released from the hydrogels was determined by liquid chromatography using the Agilent 1260 Infinity II HPLC chromatographic system. The experimental results showed that the amount of DOX loaded into the hydrogels (batch 1 - batch 3) indicated that the hydrogels loaded an amount of DOX between 1130 – 1155 ng, with ESR of 3.7 – 4.7%. In pH 6.4, the hydrogels release an amount of DOX of about 120 ng/cm^2. In the time interval between 0.5-6h, a quantity of DOX is released between 60-120 ng/cm^2.
In pH 7.4, in the same time interval, an amount of DOX between 65-150 ng/cm^2 is released, an amount of drug that is superior to that released in pH 6.4. Therefore, the designed hydrogels demonstrate DOX loading capacity and controlled drug release over a period of time between 0.5-58h. Right from the start, the hydrogels demonstrate DOX-releasing capacity, results supported by biological studies. Hydrogels loaded with DOX by diffusion show significant anti-tumor activity by reducing the number of viable melanoma-specific A375 cells.
Act. 2.2 aimed to verify the reproducibility of the obtaining method. For this purpose, 3 batches of hydrogels (100 samples/batch) were produced at intervals of 1 week, according to the optimized recipe in stage I of the project. All 3 batches presented constant values of gel fraction (ESR 1.5%) and density (ESR 0.4%). The hydrogels showed a higher absorption degree in the tumor-like environment compared to the basic environment characteristic of infected wounds. Statistically, between batches of hydrogel, ESR < 8%. We estimate that according to this quality parameter, between the batches of hydrogel from a statistical point of view, there are no differences that could critically influence the quality of the hydrogel. The hydrogel-type systems showed a predominant viscoelastic behavior (G'>>G"), and from the FTIR analysis, no variation of the characteristic absorption bands was observed between the hydrogel batches, which shows that a stable hydrogel was obtained and at the same time, the obtaining process is reproducible.
Act. 2.3 aimed to evaluate the stability of DOX during the irradiation process which was tested by liquid chromatography coupled to mass spectrometry (LC-MS) in comparison with a standard solution of non-irradiated DOX. Following irradiation, the formation of eight degradation products was observed. Of these, three are represented by the D559abc isomers observed as (–)m/z 558.16 and (+)m/z 560.17, each containing one extra oxygen atom. Another set of isomers identified is D398ab ((–)m/z 397.09 and (+)m/z 399.1), which correspond to the loss of the tetrahydropyran ring. Alternative cleavage of the ether bond in the DOX structure leads to the formation of D398ab, D412ab ((–)m/z 411.07 and (+)m/z 413.08). The difference between the two sets of isomers that lose the tetrahydropyran ring is given by the presence (D412ab) or absence (D398ab) of a carbonyl group at the position of the ether bond in the DOX structure. The last degradation product identified was D529 ((–)m/z 528.15 and (+)m/z 530.17). This suggests the loss of a carbon atom, most likely from the methoxy group, with its transformation into a hydroxyl group. Based on these results, DOX was chosen to be loaded into the hydrogel by diffusion, following the irradiation crosslinking process.
Act. 2.4 aimed to evaluate the cytotoxic and anti-proliferative potential of the drug-loaded hydrogel (DOX) capable of time-releasing an amount equivalent to the previously established IC50 dose (10 ng/ml). These results suggest using the developed hydrogel in the alternative treatment of melanoma, as a platform for targeted and controlled release of the active drug (DOX) at the cutaneous wound level.
STAGE 3 – 2024
Stage 3 of the project had as its main objective the evaluation of scientific results and the identification of novel elements that can be the subject of a national invention patent application.
The technical documentation for the patent application with the title "PROCEDURE FOR OBTAINING BIOCOMPATIBLE SEMI-SOLID HYDROGEL WITH DOXORUBICIN RELEASE AND PRO-REGENERATIVE ROLE" was developed and registered at the State Office for Inventions and Trademarks (OSIM) by the inventors: Demeter Maria (Project Leader), Călina Ion Cosmin, Scarișoreanu Anca Mariana, Cîmpean Anișoara (Project mentor) - Invention patent application A 00138/28.03.2024.